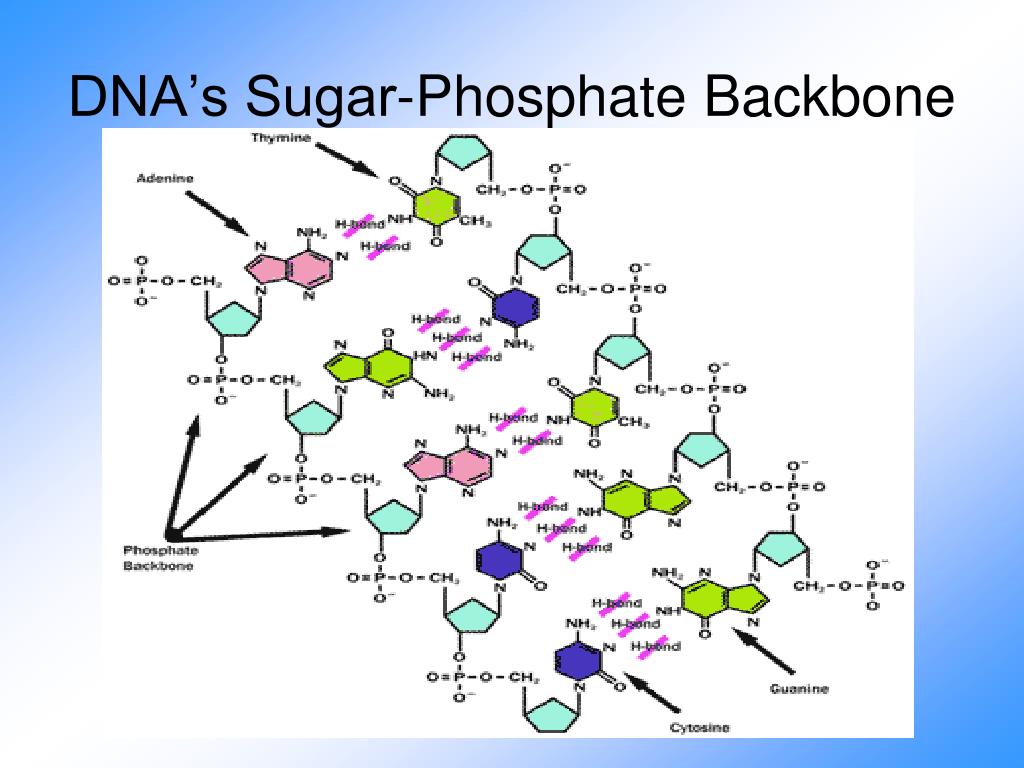
Of the major groove, as this provides more chemical information for Sequences of bases, it is not surprising that most bind to the floor Since many proteins that bind DNA recognize specific Similar considerations apply to C-GĪnd G-C base pairs. Groove ( HA- HD- HA- CH3), the edge in the minor groove ( HA- H- HA) would remain essentially the Whereas a switch to anĪ-T base pair would present a reversed edge chemistry in the major Only a HA, a hydrogen ( H), and a second HA. The minor groove edge, in contrast, presents Sterochemical environment in the major groove than in the minor groove.Īs can be observed in the T-A base pair shown, the major groove edgeĬontains a methyl group ( CH3), a hydrogen bond acceptor ( HA), a hydrogenīond donor ( HD), and a second hydrogen bond acceptor ( HA) [for simplicity, The edges of the base pairs present a more complex The distance between the sugar-phosphate backbones being much greater In B-DNA, the major groove is considerably wider than the minor groove, Although the grooves are of similar depth Of two different grooves on opposite sides of the base pairs, the Through glycosidic bonds is asymmetrical. The attachment of bases to the backbone sugars Here illustrated schematically for 1 strand of DNA. This semi-conservative mode of replication is Two daughter molecules identical to the parent, with one strand ofĮach daughter helix conserved from the parent molecule. Serves as a template upon which complementary strands are built, producing In their 1953 paper describing the first model of the DNA double helix,īase complementarity provides a means by which the genetic materialĬan replicate with fidelity. The double helix forming a sequence of bases complementary in hydrogenīonding to that of the other strand. The base pairing of opposite strands is stereochemicallyīonds are formed in A- T and G- C base pairs, respectively.īase pairing results in strand complementarity, with one strand of Is ~20 Angstroms, and the distance between base The reversed orientations of the ribose sugars on opposite strands. Is antiparallel, with one strand running 3' -> 5' and Each base forms hydrogen bonds (indicated byĭashed lines) with a base from the opposite strand. In the interior of the molecule, their planes nearly perpendicular Of each strand form the helical scaffold, with the nitrogenous bases The two strands are differentially colored to illustrate the right Structure of which was first determined by Watson and Crick. In the cell functions as a double-stranded helix of B-form DNA, the Top to bottom, how would you label the polarity of the sugar-phosphate The 5' and 3' carbons of the ribose sugars The molecule can be viewed as a series of nitrogenousīases (CPK coloration) connected through a sugar-phosphate The phosphorus is in turn linked to the 3'Ī DNA strand is thus referred to as a phospho diester linkage. Sugar by a glycosidic bond between a nitrogenĪnd the 1' carbon of the deoxyribose sugar.Įach sugar is linked to a phosphorus through an oxygen (ester bond). The carbons of the sugar are numbered 1'-5' as indicated. In the chain comprises a nitrogenous base, In the tetrameric example shown, each of the four deoxyribonucleotideĪs shown here for dC, each deoxyribonucleotide įor simplicity, hydrogen atoms are not shown. To the standard CPK coloring scheme ( C, N, O, P ). Is a short stretch of a single strand of DNA. Reset the molecule, use the reset buttons: If an "unresponsive script" error appears, click continue: However, long JSmol scripts may become unresponsive. Note: This tutorial uses JSmol and will run on a variety of platforms. To evoke renderings of the molecule that illustrate particular To the right to scroll through the text of this exhibit. This exhibit displays molecules in the left part of the screen,Īnd text that addresses structure-function relationships of
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
